Table of Contents
The conversation around Carbon Fiber Medical Devices almost always collapses into a single talking point: weight. That framing is both true and insufficient — and it leads procurement teams and product engineers to undervalue what carbon fiber actually does in a clinical engineering context.
Carbon fiber medical applications are compelling not because the material is light, but because it resolves several engineering constraints simultaneously that traditional materials address in isolation, or not at all. High structural performance at low mass. Near-complete transparency to diagnostic and therapeutic imaging radiation. Chemical stability through thousands of sterilization cycles. And geometric design freedom that isotropic materials cannot replicate at any weight.
For sourcing managers, OEM program directors, and R&D engineers evaluating a material transition, the productive question is not “how much lighter is carbon fiber?” It is: how many of these four conditions does your device application actually require? If the answer is two or more, then the material decision is largely made — what remains is execution.
This article does not argue that carbon fiber is right for every medical device. It identifies the specific product categories and performance requirements where it delivers measurable clinical and commercial value, and gives product teams a framework for making that determination before tooling commitments are made.
The Real Reason Carbon Fiber Belongs in Medical Device Engineering
Lightweight medical device materials is a specification term that appears without a performance definition attached to it. In clinical engineering, what lightweight needs to accomplish differs sharply depending on whether the device is held by a surgeon for four hours, worn daily by a mobility-impaired patient, or repositioned dozens of times per shift by a radiographer.
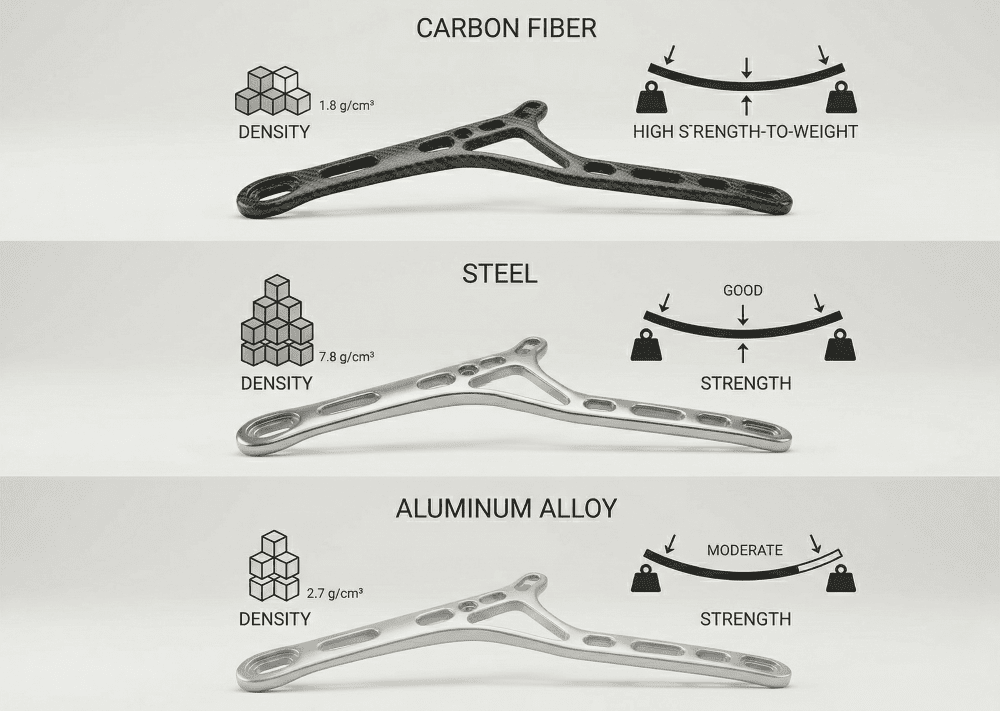
The structural case for carbon fiber starts with density. Carbon fiber composites sit at approximately 1.5–1.6 g/cm³ — roughly one-fifth the density of steel, and about 40% lower than aluminum. But density alone does not explain why carbon fiber is used in medical devices at the rates it is. The more important property is anisotropy: the stiffness of a carbon fiber component is not fixed, it is designed. By controlling the orientation and sequence of fiber layers during layup, engineers can build maximum stiffness along a primary load path while leaving the structure compliant in directions where rigidity would work against the user.
JCSPORTLINE’s finite element analysis comparing carbon fiber and metal structural components quantifies the performance gap with precision. In a direct comparison of a carbon fiber composite part against a two-piece metal assembly, the carbon fiber component achieved an axial tensile stiffness of 18,518 N/mm versus the metal’s 12,500 N/mm — a 48% increase in stiffness. Peak bending load reached 2,681 N for the composite versus 1,018 N for the metal equivalent. The composite part weighed 0.718 kg; the metal assembly weighed 1.55 kg — a 53.7% mass reduction. The composite was a single integrated part. The metal required two stamped components, four bolts, and thirty weld points. That reduction in part count and assembly complexity is directly relevant to medical device programs where assembly tolerance and repeatability affect clinical performance.
For industrial designers and product development engineers working on wearable or handheld clinical equipment, this structural efficiency translates into three concrete outcomes. Clinician fatigue from sustained tool use decreases. Patient load from worn devices — carbon fiber prosthetics, orthoses, mobility aids — decreases in ways that affect independent function over the long term. And the ability to tune stiffness directionally means the mechanical behavior of a device can be matched to how the human body actually moves, rather than constrained by what a homogeneous material happens to provide.
Radiolucency — The Property That Changes Everything in Imaging Environments
Radiolucent carbon fiber does not appear on X-ray, CT, or fluoroscopy imaging the way metal does. This single property determines material selection for an entire category of medical devices — and it is one that no lighter-gauge aluminum alloy or titanium variant can address, regardless of how thin the section is made.
Metal components in the imaging field produce high-intensity artifacts that block anatomical detail, distort radiation dose distribution in radiotherapy delivery, and obstruct the surgeon’s real-time view during image-guided interventions. The problem is not the thickness of the metal — it is the atomic density. Carbon fiber composites are largely transparent to the radiation energies used in clinical imaging, producing minimal scatter and no structural shadowing.
Carbon fiber imaging tables and radiolucent components have become standard in high-precision clinical settings for exactly this reason. Radiotherapy positioning boards, CT table extensions, surgical table overlays, and interventional radiology patient supports are all applications where the component sits directly in the imaging field during clinical use. In those environments, material selection is not a cost optimization exercise — it is a clinical requirement that removes most conventional structural materials from consideration before any other specification is evaluated.
In any device that operates within an imaging field — diagnostic, interventional, or therapeutic — radiolucency is not a feature. It is the baseline. A metal component in that position is not a cost-saving choice; it is a design error that degrades the clinical procedure it is meant to support.
For medical device innovation teams developing imaging-compatible patient platforms, the implication is straightforward: radiolucency must be specified at the concept stage, not resolved in engineering review. Retrofitting a metal-framed design to achieve imaging compatibility typically means a structural redesign, not a material swap. Carbon fiber changes the starting conditions of the design problem entirely.
→ Carbon fiber compression molding process — The manufacturing process used to produce imaging table components directly affects dimensional consistency and surface finish across production runs. Compression molding delivers the part-to-part repeatability that clinical OEM programs require.

Corrosion Resistance and Sterilization Compatibility — Built for Clinical Durability
Hospital environments are chemically aggressive on a schedule. Devices are wiped down with isopropyl alcohol, sodium hypochlorite solutions, and quaternary ammonium compounds between patient uses — sometimes dozens of times per day. Autoclave sterilization cycles add high-temperature steam pressure on top of that. The materials that perform well in those conditions are not necessarily the materials that perform well under structural load, and vice versa.
Carbon fiber composites are chemically inert to the disinfectants used in standard hospital protocols. The carbon fiber itself does not oxidize. The epoxy matrix does not react with alcohol or chlorine-based cleaning agents at the concentrations used in clinical settings. There is no surface coating to strip, no anodized layer to degrade in alkaline cleaners, and no fatigue mechanism analogous to the stress corrosion cracking that affects certain metal alloys in repeated wet-dry cycling.
For procurement officers managing device fleet replacement cycles, the durability argument for biocompatible composite materials resolves into a straightforward total-cost-of-ownership calculation. A component that maintains structural integrity and surface finish through thousands of sterilization events has a longer service life than one that degrades progressively under the same conditions. The higher unit cost of a carbon fiber component frequently compresses when amortized against a longer replacement interval — but that calculation has to be made explicitly, not assumed.
Carbon fiber sterilization and hospital durability also extends to structural components that are never cleaned directly but operate continuously in high-humidity clinical environments: imaging room support structures, rehabilitation equipment frames, mounting hardware for overhead systems. In those applications, the absence of corrosion-related maintenance requirements over the device lifetime is an operational and budget advantage that accumulates silently until a metal equivalent starts to show surface degradation.
One specification note that composite engineers and procurement teams should align on early: autoclave sterilization compatibility depends on the resin system, not just the fiber. Standard structural epoxy formulations are not always optimized for repeated high-pressure steam cycles at 134°C. Medical device programs requiring autoclave sterilization should flag that requirement at the material specification stage so the resin system and post-cure protocol are selected accordingly. This is a manageable engineering decision — it is not a reason to reject carbon fiber — but it is a decision that needs to happen at the beginning of the project, not after tooling is committed.

Where Carbon Fiber Delivers Maximum Value — Key Medical Use Cases
The four properties above do not distribute evenly across all medical product categories. The following use cases represent the segments where carbon fiber medical applications deliver the highest intersection of clinical performance and commercial differentiation.
Carbon Fiber Prosthetics and Orthopedic Supports
Carbon fiber for prosthetics and orthopedic supports is the most structurally sophisticated application of composites in routine medical use. Dynamic response prosthetic feet — the type used by active users and athletes — function because of carbon fiber’s anisotropy, not despite it. The fiber schedule in a prosthetic blade is engineered to store elastic energy during the loading phase of gait and release it at push-off, mimicking the mechanical behavior of the Achilles tendon and plantar fascia. That energy return behavior cannot be replicated in aluminum or titanium without a fundamentally different structural geometry.
Carbon fiber orthopedic devices cover a wide product range: ankle-foot orthoses, knee bracing structures, spinal orthotic shells, and prosthetic socket systems. The consistent engineering challenge across all of them is satisfying three conflicting requirements simultaneously — sufficient structural stiffness for support, low enough mass to avoid disrupting normal movement patterns, and surface geometry that conforms to variable human anatomy. Carbon fiber composite layup with custom tooling is currently the only manufacturing approach that addresses all three without material compromise.
→ Carbon fiber design services — For OEM teams developing custom prosthetic or orthotic products, the fiber schedule definition and layup engineering are as critical as the manufacturing execution. Design for composite manufacturability starts before the mold is specified.
Carbon Fiber Wheelchairs and Mobility Aids
Carbon fiber wheelchairs and mobility aids apply lightweight structural efficiency directly to a patient outcome that is both functional and measurable: how far a manual wheelchair user can travel independently, and at what physical cost.
For self-propelling users, frame mass correlates directly with the muscular effort required per stroke and the cumulative load on the shoulder and wrist joints over years of use. The Carbon Fiber Wheelchair category has expanded significantly as fabrication processes have matured. Frames, seat shells, footrests, and push-rim assemblies are all candidates for carbon fiber construction, with the aggregate mass reduction affecting daily energy expenditure in ways that standard aluminum construction cannot match without sacrificing structural rigidity.
Carbon fiber patient comfort and ergonomics in this context is not a soft product attribute — it is a clinical outcome. For OEM brands developing premium mobility products, the material also functions as a market positioning mechanism: the visible fiber texture, the reduced mass that users notice on first contact, and the engineering credibility of composite construction support pricing differentiation in a category that has historically competed almost entirely on unit cost.
→ Carbon fiber wheelchair applications — Carbon fiber wheelchair applications in development or production — specifications, use case examples, and manufacturing considerations.

Carbon Fiber Surgical Instruments
Carbon fiber surgical instruments represent a smaller but technically significant segment of carbon fiber medical applications. The engineering case centers on two properties that matter specifically in prolonged or high-precision procedures: mass and vibration damping.
Instruments used in laparoscopic surgery, microsurgery, or ophthalmic procedures are held and manipulated continuously for extended periods. Instrument mass and balance affect surgeon fatigue in the same way that any repetitive precision task does — the effect is cumulative and compounds across a full operating list. A lighter, better-balanced instrument does not change surgical skill, but it removes a physical variable that accumulates over time.
Carbon fiber’s damping characteristics — its ability to attenuate vibration within the structure — also reduce the transmission of fine hand tremor to the instrument tip. That property has driven interest in carbon fiber shafts and handles for tools where tip stability is directly related to procedural accuracy. Combined with chemical resistance to hospital-grade disinfectants, the material profile matches the requirements of reusable high-value surgical instruments well.
Imaging Tables and Radiolucent Support Components
Carbon fiber imaging tables and radiolucent components — radiotherapy couches, CT table extensions, surgical table overlays, stereotactic positioning boards — are the most established commercial application of carbon fiber in the medical sector. The structural requirement is not complex: the component must support patient weight reliably across a long service life. The imaging requirement eliminates most alternatives before structural analysis even begins.
Carbon fiber is not selected for these applications because it is the lightest or cheapest option. It is selected because it is the only structural material that simultaneously satisfies the load requirement and does not compromise the imaging procedure it is designed to support. For medical device engineers who have not worked with composites before, this use case is often the clearest entry point: the decision logic is binary, the performance requirement is unambiguous, and the commercial case is straightforward.
Customization as a Competitive Advantage — Why OEM Teams Choose Carbon Fiber for Product Differentiation
For medical device OEM decision-makers, the four functional properties of carbon fiber become commercially significant when combined with one additional characteristic: composite materials are a design medium, not just a structural material.
Compression molding, autoclave prepreg layup, and resin transfer molding all allow three-dimensional geometries that sheet metal fabrication and injection molding cannot replicate without assembly. A carbon fiber component can integrate structural ribs, mounting features, ergonomic surface contours, and a finished cosmetic surface in a single cured part. The part count reduction is real — and in medical device programs where assembly tolerance accumulates across multiple components, fewer parts means fewer sources of dimensional variation.
Custom carbon fiber components for medical OEM programs therefore compress the bill of materials while expanding the design freedom available to the product team. The same layup that establishes the stiffness distribution also defines the surface. The composite forming process that builds the structure also produces the finish — whether matte woven texture, glossy paint-ready surface, or a hybrid of both across different zones of the same part.
For medical device innovation teams developing differentiated products in markets where most competitors are working from the same aluminum extrusion and injection-molded plastic toolkit, carbon fiber provides a product identity that is genuinely difficult to replicate. The visible fiber pattern, the tactile quality of a composite surface, and the engineering narrative that comes with the material — these are not cosmetic additions. They are product differentiation mechanisms that support margin and brand positioning simultaneously.
Healthcare technology trends in rehabilitation equipment, surgical instrumentation, and patient positioning are moving toward lighter, more ergonomic, more durable products. The future of carbon fiber in healthcare is not a single application breakthrough — it is the progressive expansion of custom composite fabrication into product categories where OEM teams have historically defaulted to metal because the tooling knowledge was more accessible. That access gap is narrowing.
Carbon fiber patient comfort and ergonomics at the design level means that composite forming processes allow surface geometry to track anatomical contours with a precision that stamped metal and molded plastic cannot easily match, particularly at the production volumes typical of medical OEM programs — where tooling investment needs to stay proportional to volume, but design quality cannot be compromised.
→ Carbon fiber design services for OEM medical programs — Custom carbon fiber design services for OEM medical programs — from concept geometry through DFM review and fiber schedule specification.

When Does the Switch from Metal to Carbon Fiber Make Sense? — A Decision Framework
The most useful thing this article can do for a sourcing manager or product engineer evaluating a material transition is to be direct about when carbon fiber is not the right call — and precise about when it is.
Understanding how to evaluate carbon fiber for medical device manufacturing starts with four conditions. When a device application satisfies two or more of them, the engineering case for carbon fiber is strong enough to justify a formal feasibility assessment. When none of them apply, the economics and complexity of composite manufacturing are unlikely to produce a better outcome than conventional metal fabrication.
| Condition | What It Means in Practice |
| Weight directly affects clinical or patient outcomes | Mass reduction changes how far a mobility aid user travels independently, or how long a clinician can work without fatigue accumulation — not just a specification number. |
| Device operates in an imaging field | X-ray, CT, fluoroscopy, or radiotherapy delivery proximity eliminates most structural materials from consideration; carbon fiber’s radiolucency is non-negotiable in this context. |
| High-frequency sterilization is part of the use protocol | Hundreds or thousands of disinfection cycles per year create long-term material fatigue that carbon fiber composites avoid; replacement frequency is a total cost variable. |
| Product requires differentiated market positioning | Carbon fiber supports premium pricing and brand identity through engineering credibility and visible material quality that standard metal fabrication does not produce. |
If your project satisfies two or more of these conditions, the question is no longer whether carbon fiber is appropriate. It is how quickly you can complete a credible feasibility assessment to validate the specification and lock the cost model.
The honest counterargument: purely internal structural elements with no imaging requirement, single-use disposable devices where material cost is the dominant variable, or prototype programs with extremely limited tooling budgets may find that carbon fiber vs aluminum for medical equipment comparisons do not favor composite construction at that stage. Carbon fiber is a precision engineering tool. Using it where the precision is not required adds cost without adding value.
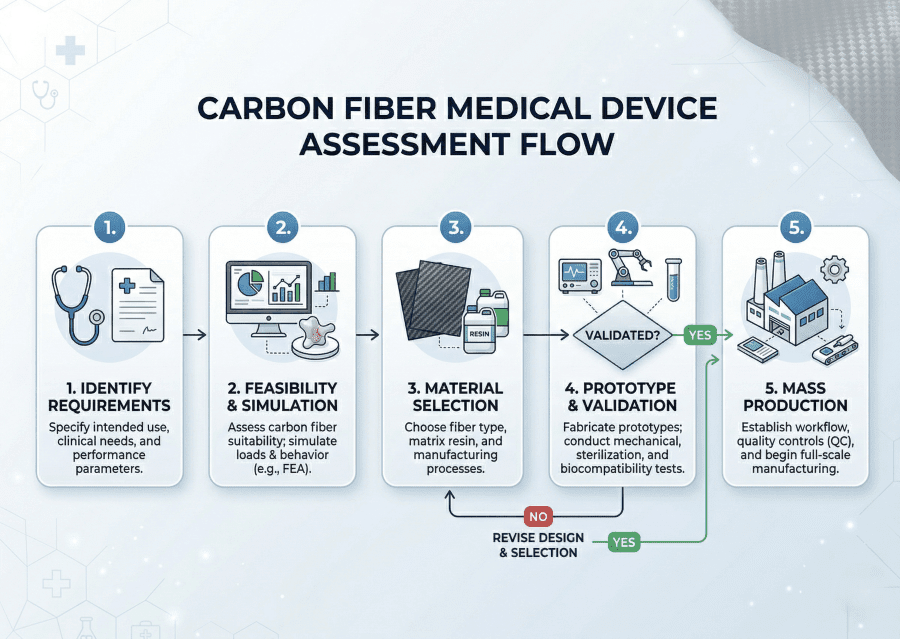
The practical first step for any team considering the transition is a technical feasibility assessment — conducted before tooling decisions are made, before BOM costs are locked, and before the design is committed to a manufacturing process. How to evaluate carbon fiber for medical device manufacturing is, at its core, a question of sequencing: get the engineering review done before the capital is committed, not after.
→ Carbon fiber project management — Project management for carbon fiber medical device programs — how JCSPORTLINE structures the feasibility-to-production workflow for OEM clients.
Conclusion
Carbon Fiber Medical Devices have a clear engineering rationale — not a material trend one.
High structural performance at low mass. No interference with diagnostic or therapeutic imaging. Chemical stability through the sterilization protocols clinical environments require. And composite forming processes that allow design teams to build complex, anatomically-matched geometries that conventional fabrication cannot produce at comparable cost and volume.
If your device program has an explicit requirement in at least two of those areas, carbon fiber is not one option among several. It is the most direct engineering path to a product that performs as the clinical application demands. The remaining question is execution — how to move from CAD to validated production without the tooling and specification risks that derail composite programs.
That execution path, from feasibility review through DFM, prototyping, and scalable manufacturing, is what JCSPORTLINE’s composite engineering team is built to deliver. If you are evaluating carbon fiber for a current or upcoming medical device program, the productive starting point is a technical feasibility conversation.
→ Carbon fiber project management — start the feasibility conversation —
Frequently Asked Questions
Is carbon fiber actually biocompatible enough for direct or near-patient contact in medical devices?
Carbon fiber is chemically inert and has been used in implant-adjacent and direct-contact applications in orthopedics and prosthetics for decades. The biocompatibility determination for a finished composite part depends on the resin system, surface treatment, and any secondary coatings — not on the carbon fiber alone. For devices with skin or tissue contact, the resin and surface specification need to be evaluated as part of the design process. That evaluation is straightforward; it is not a reason to disqualify composite construction.
How does carbon fiber actually perform through repeated autoclave sterilization — not in theory, but in service?
Performance depends on the resin system. Standard structural epoxy formulations are not always optimized for repeated 134°C steam pressure cycles. Carbon fiber composites formulated specifically for autoclave sterilization compatibility maintain structural and surface integrity through high cycle counts, but the resin selection and post-cure protocol need to be specified for that duty cycle from the start. Programs that specify autoclave compatibility after tooling is committed typically face a resin change that requires revalidation. Flag it at the concept stage.
What is the honest cost comparison between carbon fiber and aluminum for a medical device component?
Per-unit cost for carbon fiber is higher, particularly at production volumes below 1,000 units per year where tooling amortization is a significant factor. The relevant comparison is total program cost: tooling investment, assembly complexity, service life, maintenance frequency, and the commercial value of the performance differentiation. For applications where carbon fiber resolves a functional limitation that aluminum cannot address — imaging compatibility being the clearest example — the comparison is between two different products, not two versions of the same product. That distinction changes the ROI framing entirely.
Can carbon fiber components meet specific ergonomic and dimensional tolerances for medical OEM programs?
Composite tooling is designed to produce exact geometries — complex three-dimensional surfaces, integrated mounting features, anatomical contours — that would require multiple metal parts and assembly operations to approximate. Dimensional control in a carbon fiber component is a function of tooling precision and process consistency, both of which are engineering variables rather than inherent material limitations. Part-to-part repeatability in a well-controlled composite production process is sufficient for the dimensional tolerances required in medical device assembly.
What does a proper feasibility assessment for a carbon fiber medical device actually include?
A credible feasibility assessment covers: DFM review of the CAD geometry for composite manufacturability (draft angles, undercuts, fiber bridging risks at sharp radii), layup schedule definition including fiber orientation and ply count, tooling strategy and material selection based on the required production volume and curing process, an accurate BOM and unit cost model, and identification of any application-specific requirements — imaging compatibility, sterilization method, regulatory pathway — that affect material or process selection. The output is a technical roadmap that locks the cost model before tooling capital is committed. Starting with a quote before this work is done produces a number that will change.
You can also subscribe to our YouTube channel for more content like this.






